Reaction: chylomicron => TG-depleted chylomicron + 50 long-chain fatty acids + 50 diacylglycerols
- in pathway: Chylomicron remodeling
Lipoprotein lipase dimers (LPL:LPL) are tethered to heparan sulfate proteoglycans (HSPG) at endothelial cell surfaces (Fernandez-Borja et al. 1996; Peterson et al. 1992). Both syndecan 1 (Rosenberg et al. 1997) and perlecan (Goldberg 1996) HSPG molecules are capable of tethering LPL. The LPL enzyme catalyzes the hydrolysis and release of triacylglycerols (TG) associated with circulating chylomicrons to leave a CM remnant (CR). This reaction is annotated here as causing the hydrolysis and release of 50 molecules of TG. In vivo, the number is much larger, and TG depletion probably occurs in the course of multiple encounters between a chylomicron and endothelial LPL. This reaction is strongly activated by chylomicron-associated apo C-II protein both in vivo and in vitro (Jackson et al. 1986). Chylomicron-associated apoC-II protein inhibits LPL activity in vitro (Brown and Baginsky 1972), and recent studies have indicated a positive regulatory role for apoA-5 protein, though its molecular mechanism of action remains unclear (Marcais et al. 2005; Merkel and Heeren 2005). CRs can then be taken up by liver parenchymal cells in two ways; 1) directly by the LDL receptor or 2) apoE/HSPG-directed uptake by LDL receptor-related proteins.
Reaction - small molecule participants:
LCFAs [extracellular region]
DAGs [extracellular region]
Reactome.org reaction link: R-HSA-174757
======
Reaction input - small molecules:
Reaction output - small molecules:
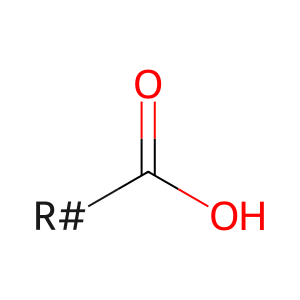
long-chain fatty acid
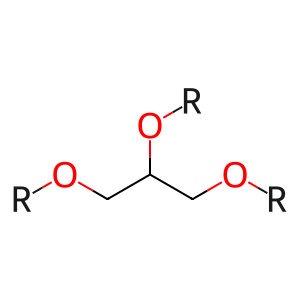
diglyceride
Reactome.org link: R-HSA-174757