Reaction: Ac-PTGS2 dimer oxidises DHA to 17-HDHA (macrophages)
- in pathway: Biosynthesis of electrophilic ω-3 PUFA oxo-derivatives
Normally, cyclooxygenases (COX) carry out stereospecific oxygenation of arachidonic acid to generate prostaglandins. When treated with aspirin (acetylsalicylic acid, ASA), dimeric cyclooxygenase 2 (COX2, PTGS2 dimer) can be acetylated. ASA covalently modifies PTGS2 by acetylating a serine residue at position 530 within the cyclooxygenase active site (Lucido et al. 2016). Acetylated PTGS2 dimer (Ac-PTGS2 dimer) changes the oxygenation stereospecificity towards its substrates, perhaps by steric shielding effects (Tosco 2013), producing a shift in lipid mediator production. Ac-PTGS2 dimer expressed in macrophages can be acetylated by ASA, which enables this form to mediate the biosynthesis of precursors of endogenous anti-inflammatory mediators. Ac-PTGS2 dimer is able to incorporate molecular oxygen into ω-3 fatty acid docosahexaenoic acid (DHA), to form 17-hydroxy-docosahexaenoic acid (17-HDHA) (Serhan et al. 2002, Groeger et al. 2010).
Reaction - small molecule participants:
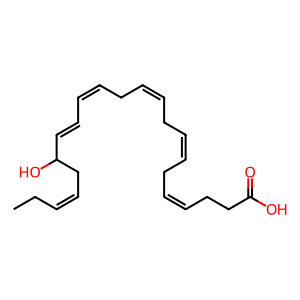
17-HDHA [cytosol]
O2 [cytosol]
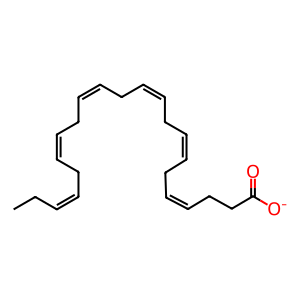
DHA [cytosol]
Reactome.org reaction link: R-HSA-9027627
======
Reaction input - small molecules:
dioxygen
(4Z,7Z,10Z,13Z,16Z,19Z)-docosahexaenoate
Reaction output - small molecules:
(4Z,7Z,10Z,13Z,15E,19Z)-17-hydroxydocosahexaenoic acid
Reactome.org link: R-HSA-9027627