Reaction: SPRY2 is phosphorylated by phosphorylated MNK1
- in pathway: Spry regulation of FGF signaling
In humans, the phosphorylated MNK1 kinase phosphorylates the adaptor protein Sprouty2 on Ser112 and Ser121, and also at some other serine and threonine residues. MNK1 appears not to form a complex with Sprouty2. Some of these (including the two main sites mentioned above) conform to the serine-containing consensus sites for phosphorylation by MNK1 kinase (K/R-X-X-S, R-X-S). It appears that serine phosphorylation is required to protect Sprouty2 from degradation.
In the absence of serine phosphorylation, phosphorylation of Tyr55 and subsequent binding to E3 ubiquitin ligase, CBL, is enhanced. Serine phosphorylation of Sprouty2 appears to stabilise the protein by interfering with its potential phosphorylation of Tyr55 (Sprouty2 appears to be a poor substrate for c-Src kinase) in response to growth factor stimulation.
In the absence of serine phosphorylation, phosphorylation of Tyr55 and subsequent binding to E3 ubiquitin ligase, CBL, is enhanced. Serine phosphorylation of Sprouty2 appears to stabilise the protein by interfering with its potential phosphorylation of Tyr55 (Sprouty2 appears to be a poor substrate for c-Src kinase) in response to growth factor stimulation.
Reaction - small molecule participants:
ADP [cytosol]
ATP [cytosol]
Reactome.org reaction link: R-HSA-934559
======
Reaction input - small molecules:
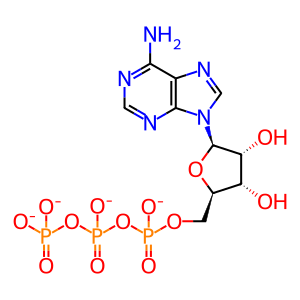
ATP(4-)
Reaction output - small molecules:
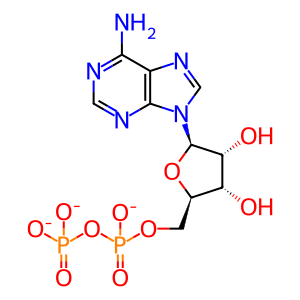
ADP(3-)
Reactome.org link: R-HSA-934559